Is Hypochlorous Acid a Strong Acid
We hear this question almost every week: Is hypochlorous acid a strong acid?
We smile because the answer surprises many people. It sounds scary, yet it is gentle. It sounds harsh, yet it protects skin and surfaces. Let’s walk through the science together in plain English.
At Shandong Shine Health Co., Ltd., we work with this solution every day. We test it. We use it. We trust it. So we want to explain the truth in a friendly way.
What Does “Strong Acid” Really Mean?
Before we judge any chemical, we must understand the term strongly acid. In chemistry, a strong acid releases many hydrogen ions in water. These acids break apart fully.
Examples include sulfuric acid and hydrochloric acid. They burn skin fast. They react with metals. They demand careful handling.
However, a weak acid releases only a small amount of hydrogen ions. It stays mostly intact in water. Hypochlorous acid falls into this gentle group.
So the answer starts here: hypochlorous acid hocl is a weak acid.
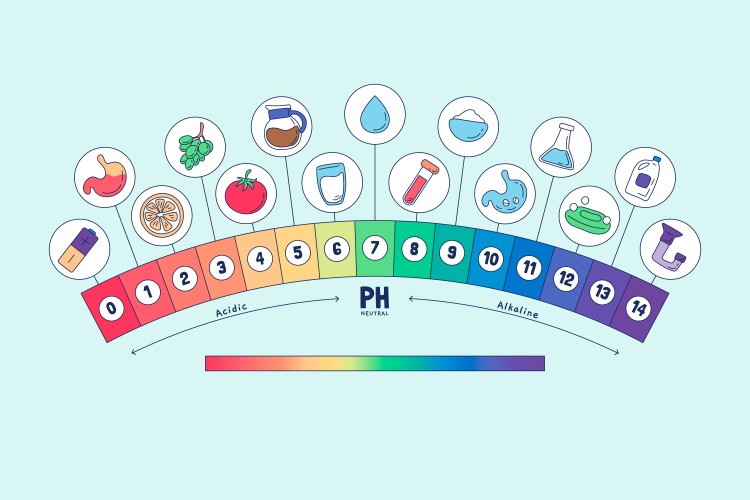
Understanding the pH Scale
The pH scale shows acidity or alkalinity. The scale ranges from 0 to 14, or written as 0 to 14.
0–3 → strongly acid
4–6 → slightly acid
7 → neutral
8–10 → mild alkaline
11–14 → high ph alkaline
Each step changes strength by 10×. Yes, chemistry loves drama.
Where Does HOCl Sit on the Scale?
HOCl works best at pH 5–6.5. This range is mildly acidic. It feels close to natural skin pH.
So even though it sounds like acid, it behaves gently. It cleans. It disinfects. It rarely irritates.
That balance makes it useful in:
wound care
food surface cleaning
home disinfection
hospital sanitation
Not bad for a “scary acid,” right?
Why HOCl Is a Weak Acid
Hypochlorous acid forms when chlorine dissolves in water. The reaction creates HOCl and a hypochlorite ion.
In simple terms:
chlorine + salt and water → HOCl + OCl⁻
Because it does not split fully, HOCl stays mild. Chemists call this partial dissociation.
The Role of Sodium Hypochlorite
Many people know bleach, or sodium hypochlorite. That solution has a high ph. It feels harsh. It smells strong.
Inside bleach, HOCl changes into the hypochlorite ion quickly. That shift lowers disinfecting power. It also raises irritation.
So we control ph levels carefully. We keep HOCl slightly acid to keep it strong against germs but gentle to skin.
From Sodium Chloride to HOCl
An electrolyzed system uses sodium chloride and water. It passes electricity through the mix. Then it creates HOCl safely.
That means we can make fresh disinfectant on site. No heavy chemical transport. No storage risk.
This process produces solutions around 200 PPM for many cleaning jobs. It keeps safety high and smell low.
Why Nature Uses HOCl
Here is a cool twist. Our white blood cells produce hypochlorous acid.
When germs invade, our immune system releases HOCl. It kills bacteria fast. It protects wounds.
Nature chose HOCl because it works. And it works gently inside our bodies.
That fact always amazes our customers. Chemistry can be kind.
HOCl in Wound Care
Doctors use HOCl in acidic water sprays. They make clean cuts without burning skin.
Because HOCl stays mild, it reduces pain. It supports healing. It lowers infection risk.
We often hear from nurses. They love it. Patients smile more. That alone makes our work worth it.
Comparing Strong Acid vs HOCl
Let’s compare side by side.
Feature | Strong Acid | HOCl |
pH | Often 0–2 | 5–6.5 |
Skin Safety | Dangerous | Safe in correct range |
Dissociation | Complete | Partial |
Use | Industry | Cleaning & care |
Strong acids destroy. HOCl disinfects gently.
So again, HOCl is not a strong acid. It is a slightly acid weak acid.
Acidic Solutions vs Safety
Some people fear all acidic solutions. That fear makes sense. Many acids burn.
But remember:
Lemon juice is acidic.
Vinegar is acidic.
Yogurt is acidic.
Acid does not always mean danger. It depends on strength.
HOCl lives in the safe zone.
Why pH Matters for Disinfection
HOCl changes form across a wide range of pH. At low pH, HOCl dominates. At high pH, hypochlorite ion dominates.
HOCl kills microbes much faster. Hypochlorite works more slowly.
So we control pH like a chef controls heat. Too high, and the power drops. Too low, and stability drops.
The sweet spot is pH 5–6.5.
Measuring the Right Concentration
We also check the concentration in PPM.
Common examples:
20–50 PPM → skin and toys
50–100 PPM → food contact surface
100–200 PPM → general disinfection
200+ PPM → heavy cleaning
Even at 200 PPM, HOCl stays gentle compared to bleach.
How We Produce HOCl Safely
We use a modern HOCl Generator to mix salt and water with electricity. The system creates stable HOCl on demand.
This method avoids shipping chemicals. It reduces cost. It improves freshness.
We also test every batch. We check pH. We check PPM. We check stability.
Quality matters to us because safety matters to families.
Tips for Using HOCl at Home
If you use HOCl, keep it simple.
Store in opaque bottles
Avoid sunlight
Use within shelf life
Check pH if possible
Shake gently before use
Fresh solution works best. The old solution loses strength.
And please, never mix with other cleaners. Chemistry can get jealous.
Common Myths About Hypochlorous Acid
We hear funny myths at trade shows. Let’s clear a few.
Myth 1: It is a strong acid.
Truth: It is a weak acid.
Myth 2: It is just bleach.
Truth: Bleach has a high pH and behaves differently.
Myth 3: It harms skin.
Truth: Correct pH solutions support wound care.
Myth 4: It smells like chlorine.
Truth: Fresh HOCl has a mild pool scent at most.
Science beats rumors every time.
The “Small Amount” Rule
More is not always better. A small amount of HOCl often kills germs quickly.
Over-concentration wastes the solution. It does not improve safety.
So we always match PPM to the task. Smart chemistry saves money.
Why We Love Working With HOCl
We admit it. We feel proud when we see clean hospitals. We smile when farmers protect animals safely. We relax when parents clean toys gently.
HOCl gives us that feeling. It solves problems without harsh chemicals.
Yes, it is chemistry. But it feels human.
Final Answer: Is Hypochlorous Acid a Strong Acid?
No. Hypochlorous acid is a weak acid. It works at pH 5–6.5. It sits far from strong acid danger.
It forms from salt and water. It appears in our immune system. It cleans surfaces safely.
So next time someone fears the word “acid,” tell them this story. Science can be gentle. And HOCl proves it.
